The computer vision leader offers controlled release cloud-based software to speed up innovation for life-enhancing drugs and medical devices
Palo Alto, CA, May 2, 2023 — Landing AI, the leading computer vision cloud platform, today announced the validation-ready version of its LandingLens platform. This new platform version is targeted at highly regulated industries, such as life sciences, drug and medical device makers, so they can speed up innovative solutions that improve the lives of everyone around the world.
Companies regulated by the Food and Drug Administration (FDA) need to validate the tools that they use, everything from filling pill vials to manufacturing medical devices. This validation process can take months and require future updates as the tools change.
With the validation-ready version of LandingLens, companies can now streamline and speed-up the validation process from three months to just one or two weeks, as well as improve the reliability and quality by following a standard process. This reduces the risk of running afoul of FDA regulations, which can cost companies millions of dollars. LandingLens is the only deep learning cloud-based platform that supports validation.
“AI, starting with computer vision, is a game-changing tool for medical device, pharmaceutical and life sciences industries, and we’ll see rapid innovation in the coming years,” said Andrew Ng, Landing AI CEO and noted AI pioneer. “This new deployment of LandingLens will make that technology available to more companies for more use cases while also improving collaboration and accessibility via the cloud.”
Leading life science and medical device companies are already using LandingLens to classify pills, inspect vials, conduct quality and assurance for packaging, or find and eradicate defects in medical device components, down to the integrity of soldering joints. With Landing AI’s computer vision platform, these visual inspections happen faster and with greater reliability than if done manually. LandingLens makes it easy to acquire and label data, ensure data quality, and train AI models. The platform is fast and intuitive, enabling teams of all skill levels to build deep learning models.
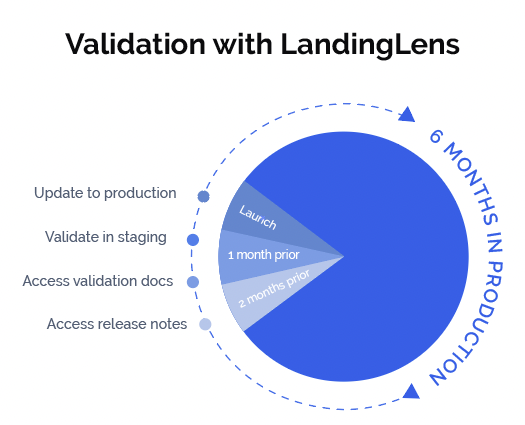
The validation-ready version of LandingLens is on a controlled release cycle, and will update every six months. In the fifth month, companies will learn of upcoming features and be able to document, test and validate them within a staging environment prior to the new release being pushed to a production environment. Because it is cloud-based, the validation-ready version of LandingLens works with any hardware. Companies can access other benefits of the cloud, such as scaling up or down based on their needs.
Landing AI is partnering with Verista, a leader in validation and vision automation, to produce documentation and templates that simplify the review process for companies deploying LandingLens.
Additional Resources
- Accelerate FDA Compliance for Your Computer Vision Systems Webinar
- Streamline Compliance: Expedite FDA Validation for Computer Vision Systems White Paper
- Validation with LandingLens Blog & FAQ
About Landing AI
Landing AI™ is democratizing the creation of AI in computer vision projects. Even with limited data sets, companies realize the value of AI and move AI projects from proof-of-concept to production. Guided by a data-centric AI approach, Landing AI’s flagship product is LandingLens™, a computer vision cloud platform that enables users to build, iterate and deploy computer vision solutions quickly and easily. With data quality being key to the success of production AI systems, LandingLens™ enables users to achieve optimal data accuracy and consistency. Founded by Andrew Ng, co-founder of Coursera, founding lead of Google Brain, and former chief scientist of Baidu, Landing AI is uniquely positioned to lead the development of AI from a technology that benefits a few to a technology that benefits all. Get started for free: www.landing.ai