Document Processing in Biotechnology
Biotechnology companies process enormous volumes of documents across every stage of drug development — IND applications, batch manufacturing records, clinical study reports, deviation reports, CMC data packages, protocol amendments, the list goes on.
Slow document review delays clinical milestones and regulatory submissions, while errors in manufacturing records introduce quality risks that stall batch release and erode program timelines.
LandingAI transforms documents into highly accurate, verifiable, structured data so teams can reliably automate document-intensive workflows.
Why Agentic Document Extraction for
Biotechnology
Accelerate Program Timelines
Extracting data from regulatory submissions, study reports, and CMC packages in hours rather than days compresses the review cycles that determine when a program advances to the next stage.
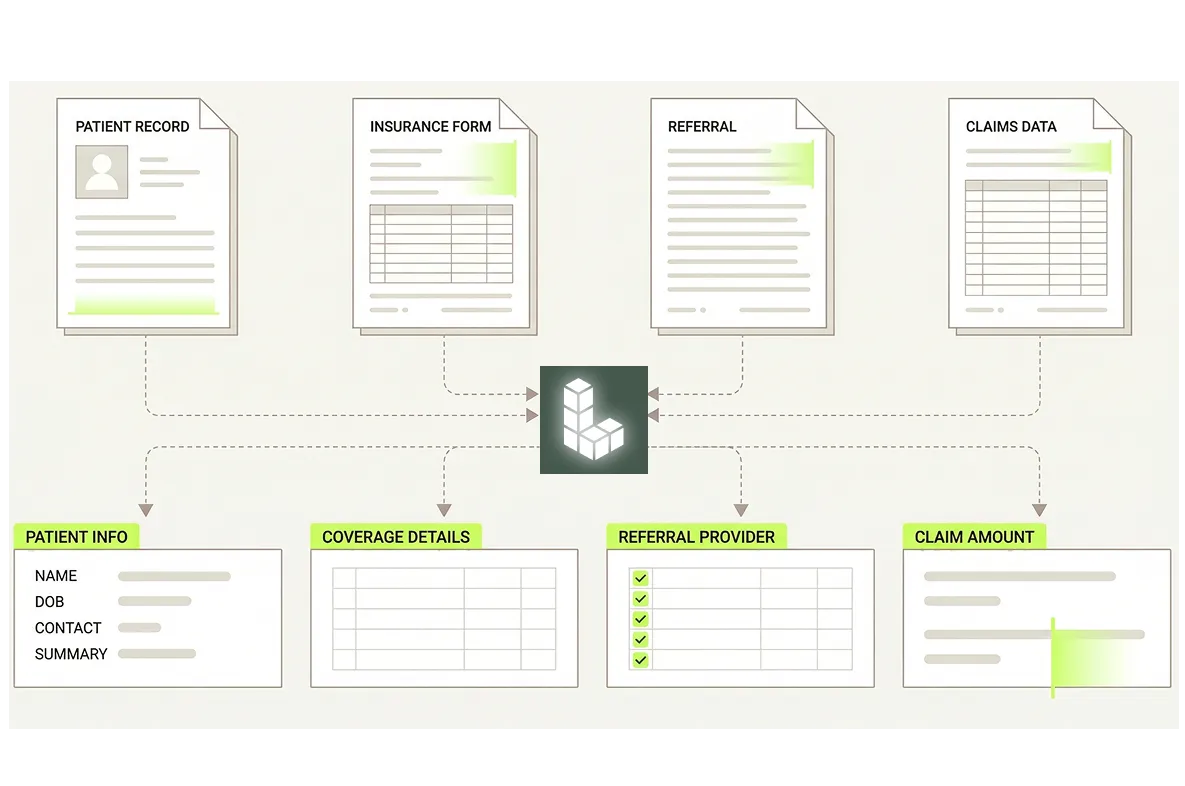
Audit-Ready Traceability by Default
Every extracted value is grounded to its precise location in the source document, giving quality and regulatory affairs teams a defensible, traceable record without manual annotation or after-the-fact reconstruction.
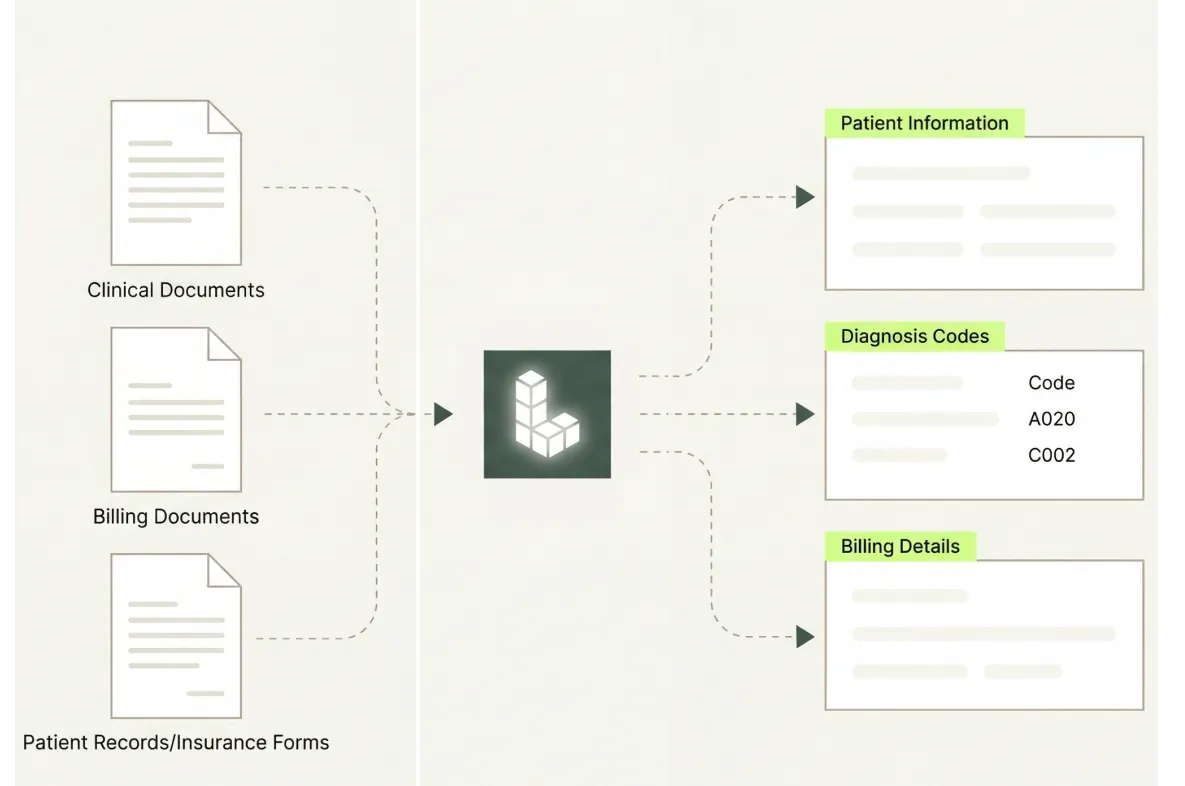
Scale Without Adding Review Headcount
Document volume grows with every clinical phase and manufacturing campaign, but extraction throughput scales through the API — so teams handle more documents without proportional growth in review staff.
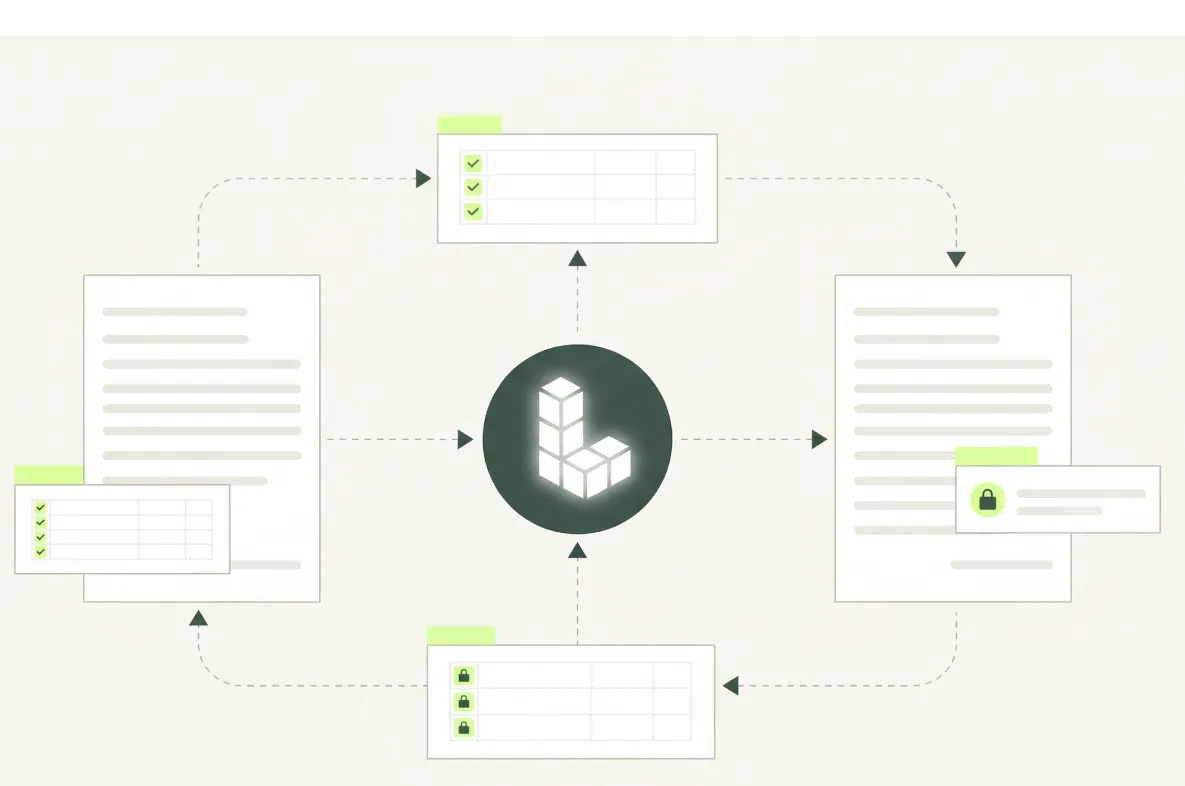
Built for Complex Biotechnology Documents
Intelligent document processing across drug discovery and development, biologics and biosimilar manufacturing, cell and gene therapy, and contract development and manufacturing organizations (CDMOs) is extremely difficult due to the sheer diversity of document types, the inconsistent layouts and the domain expertise required. Then add multiple languages, handwriting, photographs, scans and faxes to the complexity.
Accurate parsing of dense tables that span multiple pages and contain merged cells.
Single pipeline for image, slide, document, and spreadsheet file types with 1000+ pages.
Strong recognition of character-based languages, handwriting, checkboxes, stamps and signatures.
Schema-driven field extraction with visual grounding traceable to the original document.
Use cases

Regulatory Submission Assembly
Extract, organize, and verify data from IND applications, BLA modules, CMC packages, clinical study reports, and nonclinical summaries to accelerate dossier preparation and review.
Shorten submission preparation cycles to advance programs to approval faster
Reduce the manual effort required to compile and cross-check dossier sections
Ensure complete, traceable extraction that supports regulatory review and audit response

Batch Record Review
Extract and validate data from executed batch manufacturing records, deviation reports, environmental monitoring logs, and certificate of analysis documents to support batch disposition decisions.
Accelerate batch release by surfacing exceptions and deviations without manual page review
Reduce quality review labor costs on high-volume manufacturing campaigns
Maintain a complete, defensible quality record that withstands GMP inspection scrutiny

Clinical Trial Data Extraction
Extract patient data, adverse event records, investigator signatures, and protocol deviation entries from case report forms, site monitoring reports, and source documents across multi-site studies.
Compress database lock timelines to shorten the path from last patient visit to regulatory submission
Cut source data verification costs by automating reconciliation across site documents
Reduce transcription errors that generate data queries and delay study closeout
Trusted for Document-Heavy Biotechnology Workflows
Agentic Document Extraction enables biotechnology companies to automate document-intensive processes that traditionally require manual review.
Agentic Document Extraction has proven to be both accurate and easy to use. We are building on that foundation to deliver reliable, transparent, and scalable automation that our customers can validate and trust.”
View case study →
ADE has significantly outperformed other document extractors we’ve used. It has helped us build an Agentic RAG answer engine, based on unique healthcare institutional content, to offer instant, validated support to medical professionals at the point of care.”
View case study →Our Plan Review Agent has a lot of complicated components under the hood: traversing building code knowledge graphs, reasoning across disciplines and sheets, assessing issues informed by historical projects. None of it works if we can’t trust what came off the page. ADE gave us a reliable foundation, so our team could focus on incorporating our team’s expertise into our compliance reasoning system.”
View case study →
Trust is the product. Accuracy alone isn’t enough at enterprise scale—what matters is provenance, traceability, and control. LandingAI gives us confidence that every extracted value can be traced back to its source, audited, and defended. That’s what makes it deployable in regulated, real-world environments.”
View case study →
Agentic Document Extraction has proven to be both accurate and easy to use. We are building on that foundation to deliver reliable, transparent, and scalable automation that our customers can validate and trust.”
View case study →
ADE has significantly outperformed other document extractors we’ve used. It has helped us build an Agentic RAG answer engine, based on unique healthcare institutional content, to offer instant, validated support to medical professionals at the point of care.”
View case study →Our Plan Review Agent has a lot of complicated components under the hood: traversing building code knowledge graphs, reasoning across disciplines and sheets, assessing issues informed by historical projects. None of it works if we can’t trust what came off the page. ADE gave us a reliable foundation, so our team could focus on incorporating our team’s expertise into our compliance reasoning system.”
View case study →
Trust is the product. Accuracy alone isn’t enough at enterprise scale—what matters is provenance, traceability, and control. LandingAI gives us confidence that every extracted value can be traced back to its source, audited, and defended. That’s what makes it deployable in regulated, real-world environments.”
View case study →

