Document Processing in Containers & Packaging
Containers and packaging companies process millions of documents every day—food contact material declarations, substance migration test reports, artwork approval records, customer packaging specifications, EU Declarations of Compliance, extended producer responsibility filings, Unique Device Identifier submissions, and returnable container tracking records, the list goes on.
Compliance documentation gaps trigger regulatory holds, and approval bottlenecks push back launch dates that cannot be recovered.
LandingAI transforms documents into highly accurate, verifiable, structured data so teams can reliably automate document-intensive workflows.
Why Agentic Document Extraction for
Containers & Packaging
Traceable Outputs for Compliance Audits
Every extracted value from substance migration test reports, Declaration of Compliance documents, and UDI label records is grounded to a precise location in the source document, giving regulatory and quality teams the audit trail that FDA inspections, EU food contact enforcement, and EPR compliance reviews require.
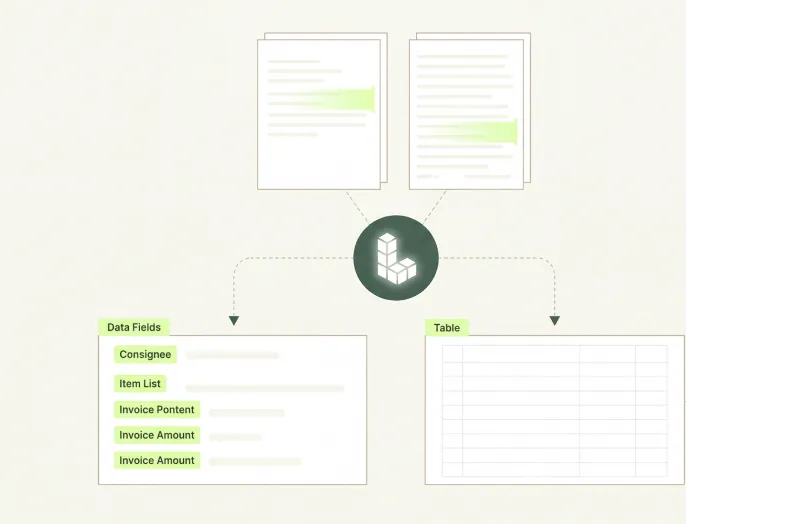
Accurate across Packaging Document Formats
Packaging documents span handwritten batch production records, dense tabular substance migration test reports, multilingual EU compliance declarations, and multi-stakeholder artwork approval files; agentic extraction handles all of them without document-specific configuration.
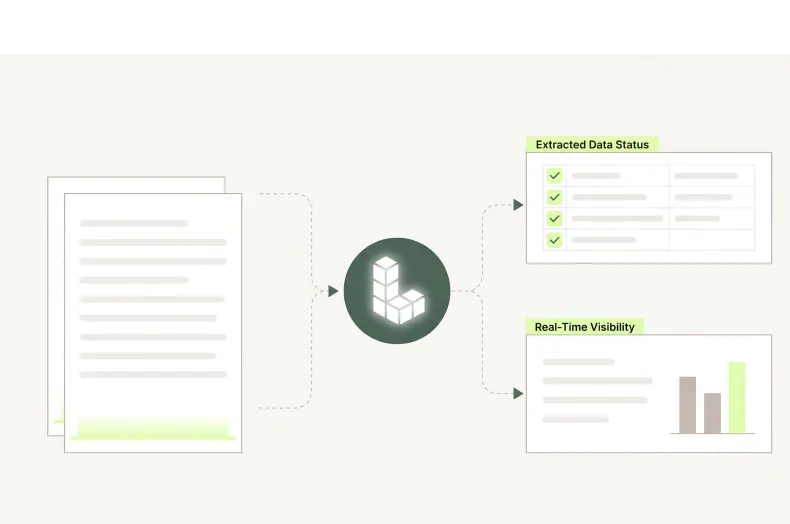
Speed That Protects Launch Windows
Compressing document processing cycle times across artwork approval, food contact certification, and EPR compliance filing reduces the administrative delays that push back retail window dates, delay regulatory clearances, and expose brands to sustainability reporting penalties.
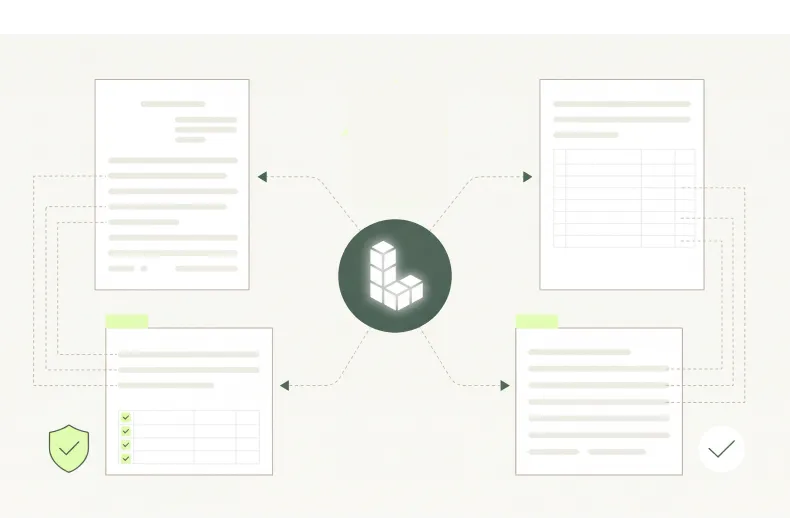
Built for Complex Packaging Documents
Intelligent document processing across rigid packaging manufacturing, flexible packaging manufacturing, pharmaceutical and medical device packaging, and contract packaging and co-packing operations is extremely difficult due to the sheer diversity of document types, the inconsistent layouts and the domain expertise required. Then add multiple languages, handwriting, photographs, scans and faxes to the complexity.
Accurate parsing of dense tables that span multiple pages and contain merged cells.
Single pipeline for image, slide, document, and spreadsheet file types with 1000+ pages.
Strong recognition of character-based languages, handwriting, checkboxes, stamps and signatures.
Schema-driven field extraction with visual grounding traceable to the original document.
Use cases

Food Contact Material Compliance
Extract authorized substance lists, migration limit test results, compliance statements, and material composition data from substance migration test reports, EU Declarations of Compliance, FDA 21 CFR Part 177 certifications, and Food Contact Notification submissions to support food safety regulatory approval and ongoing compliance across packaging material portfolios.
Accelerate regulatory clearance for new food contact materials to protect product launch timelines and retail window commitments
Cut regulatory affairs staff hours spent manually extracting and cross-referencing test results across migration studies and compliance declarations
Maintain the complete, traceable documentation chain that FDA inspections, EU food contact enforcement actions, and customer regulatory audits require

Artwork Approval & Specification Management
Extract dimensional specifications, print requirements, color codes, regulatory label elements, and stakeholder approval records from customer packaging specification sheets, artwork proof files, print specification documents, and approval feedback records to accelerate multi-stakeholder review cycles and ensure specifications are accurately captured before production release.
Compress artwork approval cycle times to protect retail window dates and reduce missed launch opportunities tied to specification delays
Reduce brand, regulatory, and supply chain staff hours spent manually routing, consolidating, and reconciling feedback across artwork revision cycles
Minimize production rework and compliance violations from specification errors with traceable, structured approval records for every SKU

EPR & Sustainability Compliance Reporting
Extract material type, weight, recycled content percentages, recyclability grades, and UPC-level packaging data from supplier material certificates, recyclability certification reports, post-consumer recycled content declarations, and product specification sheets to support state EPR compliance filings and EU Packaging and Packaging Waste Regulation reporting obligations.
Accelerate EPR filing preparation to meet state submission deadlines and minimize late-filing penalty exposure across active EPR programs
Cut sustainability staff hours spent manually aggregating material composition data from multiple packaging suppliers and SKU-level specifications
Maintain auditable material sourcing records that withstand PRO compliance reviews, state regulator audits, and PPWR Digital Product Passport requirements
Trusted for Document-Heavy Packaging Workflows
Agentic Document Extraction enables containers and packaging companies to automate document-intensive processes that traditionally require manual review.
Agentic Document Extraction has proven to be both accurate and easy to use. We are building on that foundation to deliver reliable, transparent, and scalable automation that our customers can validate and trust.”
View case study →
ADE has significantly outperformed other document extractors we’ve used. It has helped us build an Agentic RAG answer engine, based on unique healthcare institutional content, to offer instant, validated support to medical professionals at the point of care.”
View case study →Our Plan Review Agent has a lot of complicated components under the hood: traversing building code knowledge graphs, reasoning across disciplines and sheets, assessing issues informed by historical projects. None of it works if we can’t trust what came off the page. ADE gave us a reliable foundation, so our team could focus on incorporating our team’s expertise into our compliance reasoning system.”
View case study →
Trust is the product. Accuracy alone isn’t enough at enterprise scale—what matters is provenance, traceability, and control. LandingAI gives us confidence that every extracted value can be traced back to its source, audited, and defended. That’s what makes it deployable in regulated, real-world environments.”
View case study →
Agentic Document Extraction has proven to be both accurate and easy to use. We are building on that foundation to deliver reliable, transparent, and scalable automation that our customers can validate and trust.”
View case study →
ADE has significantly outperformed other document extractors we’ve used. It has helped us build an Agentic RAG answer engine, based on unique healthcare institutional content, to offer instant, validated support to medical professionals at the point of care.”
View case study →Our Plan Review Agent has a lot of complicated components under the hood: traversing building code knowledge graphs, reasoning across disciplines and sheets, assessing issues informed by historical projects. None of it works if we can’t trust what came off the page. ADE gave us a reliable foundation, so our team could focus on incorporating our team’s expertise into our compliance reasoning system.”
View case study →
Trust is the product. Accuracy alone isn’t enough at enterprise scale—what matters is provenance, traceability, and control. LandingAI gives us confidence that every extracted value can be traced back to its source, audited, and defended. That’s what makes it deployable in regulated, real-world environments.”
View case study →

