Pharmaceutical
Defective parts or products can lead to serious liability issues,
shorter product life spans, safety concerns, recalls, and additional costs.
“The platform allows us to generate accurate and consistent datasets that we can iterate over time to continuously improve our existing AI systems.”
— Ligand Pharmaceuticals
Applications
Machine vision systems and AI help medical and pharmaceutical manufacturers with quality control via automated inspection. LandingLens offers a standardized deep learning solution platform that improves inspection accuracy, reduces development time, and allows companies to easily scale projects to multiple facilities. Learn more about AI in pharmaceuticals below.
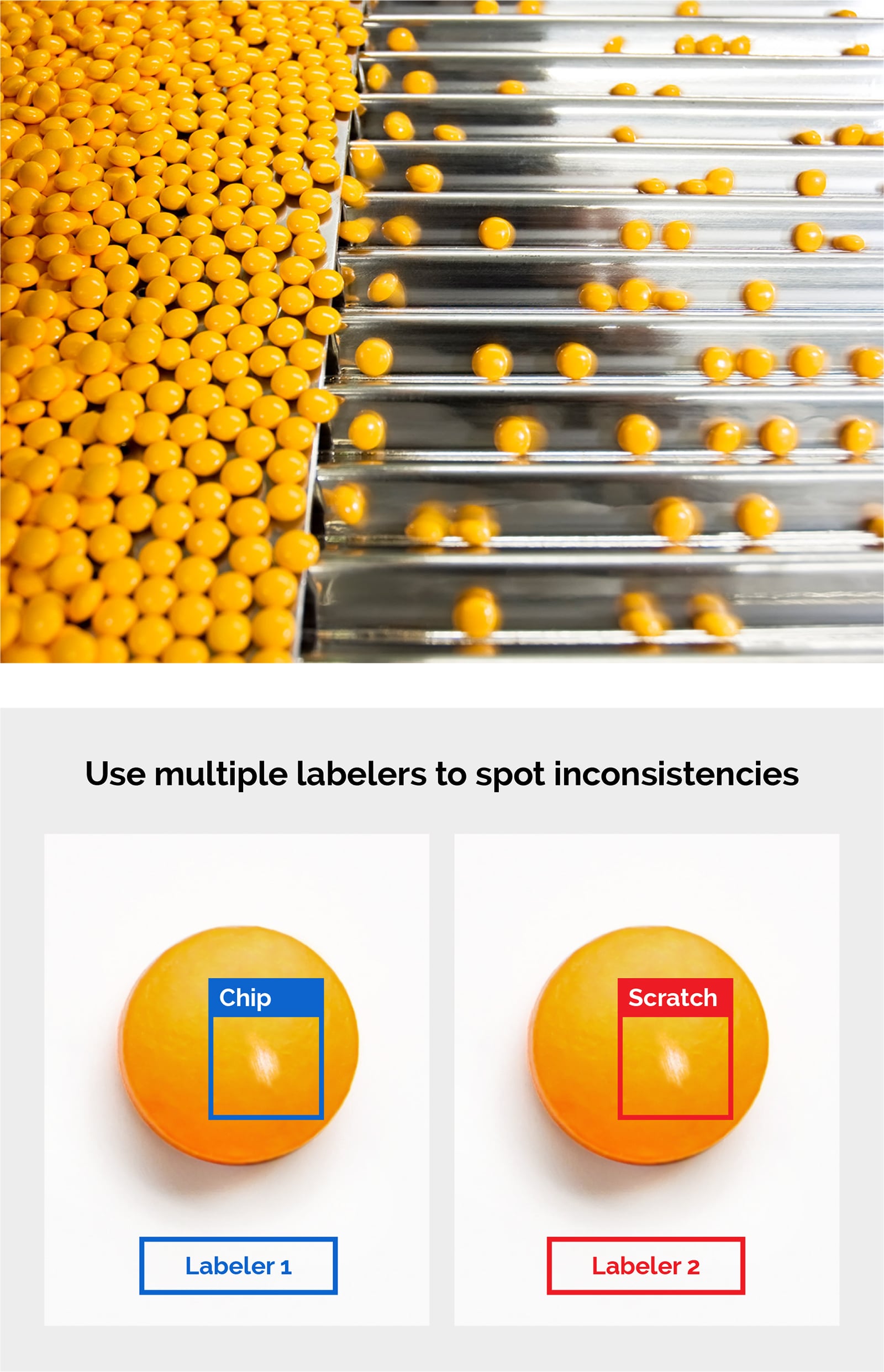
- Pill InspectionPrior to being placed into bottles and other containers, tablets and capsules must be inspected for defects and abnormalities such as cross-contamination, broken pills, and incorrect pills. Machine vision systems have long helped with such visual inspection tasks, but deep learning software can improve inspection accuracy of such systems. It can inspect large numbers of products and devices, since it is trained on images of defective and non-defective material.
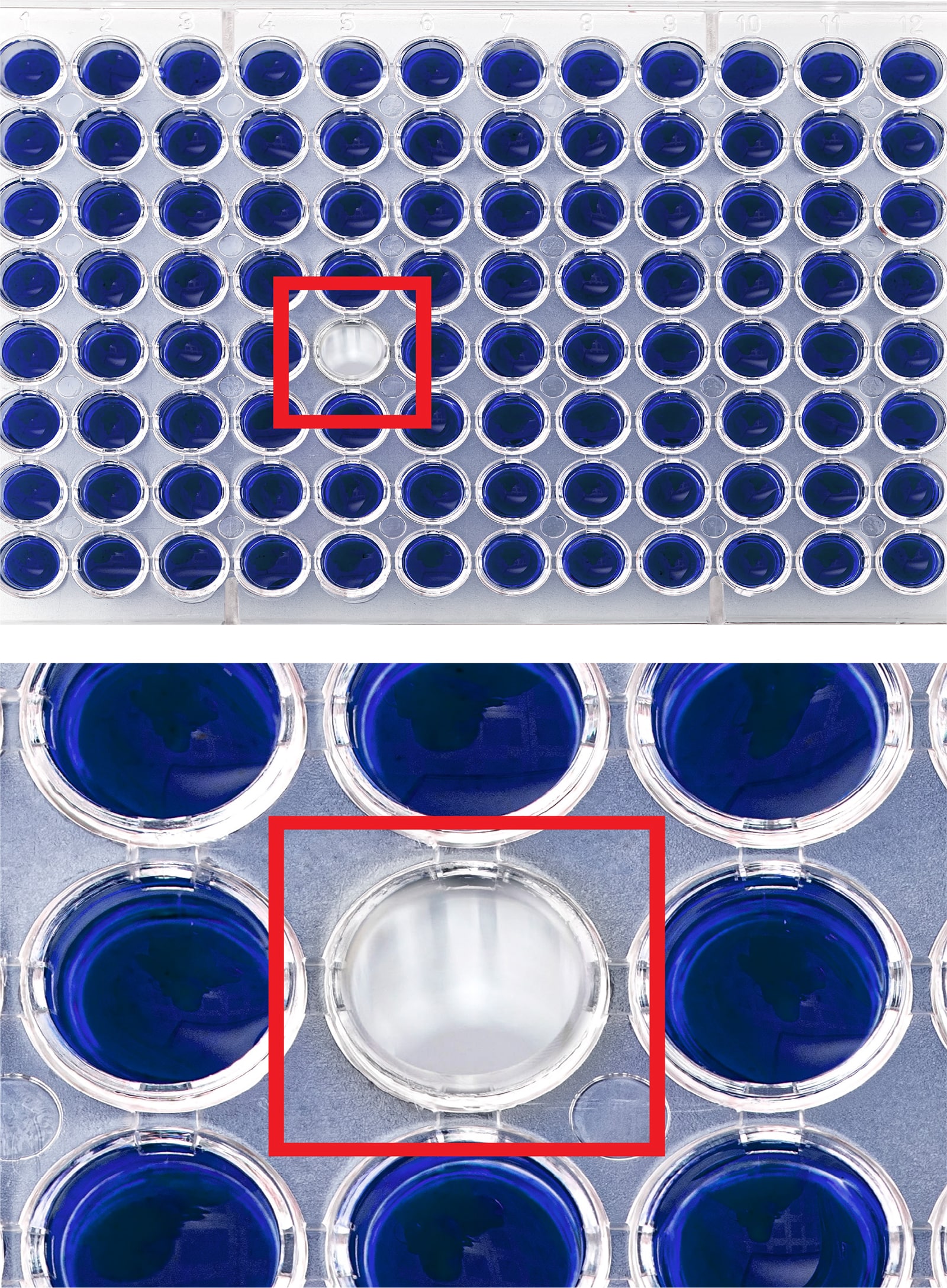
- Vial CountingMachine vision technology can automate the process of vial counting, saving time and removing the risks involved with human error, such as misplaced or missing vials and subsequent recalls. Machine vision systems have visual counting capabilities, but deep learning software such as LandingLens provides a secondary layer of protection to ensure the correct number of vials and that they do not have defects.
- Medical Device Inspection (Nano-Scale)While several inspection tasks in medical and pharmaceutical settings can be done manually, the inspection of nano-scale components is not one of them. Manufacturers must deploy imaging technology to inspect for defects and abnormalities, oftentimes down to the micron level. This involves manual inspection with a microscope, which includes multiple steps and can be susceptible to human errors and inconsistency. Deep learning software can automatically process microscopy images and classify defects or irregularities.
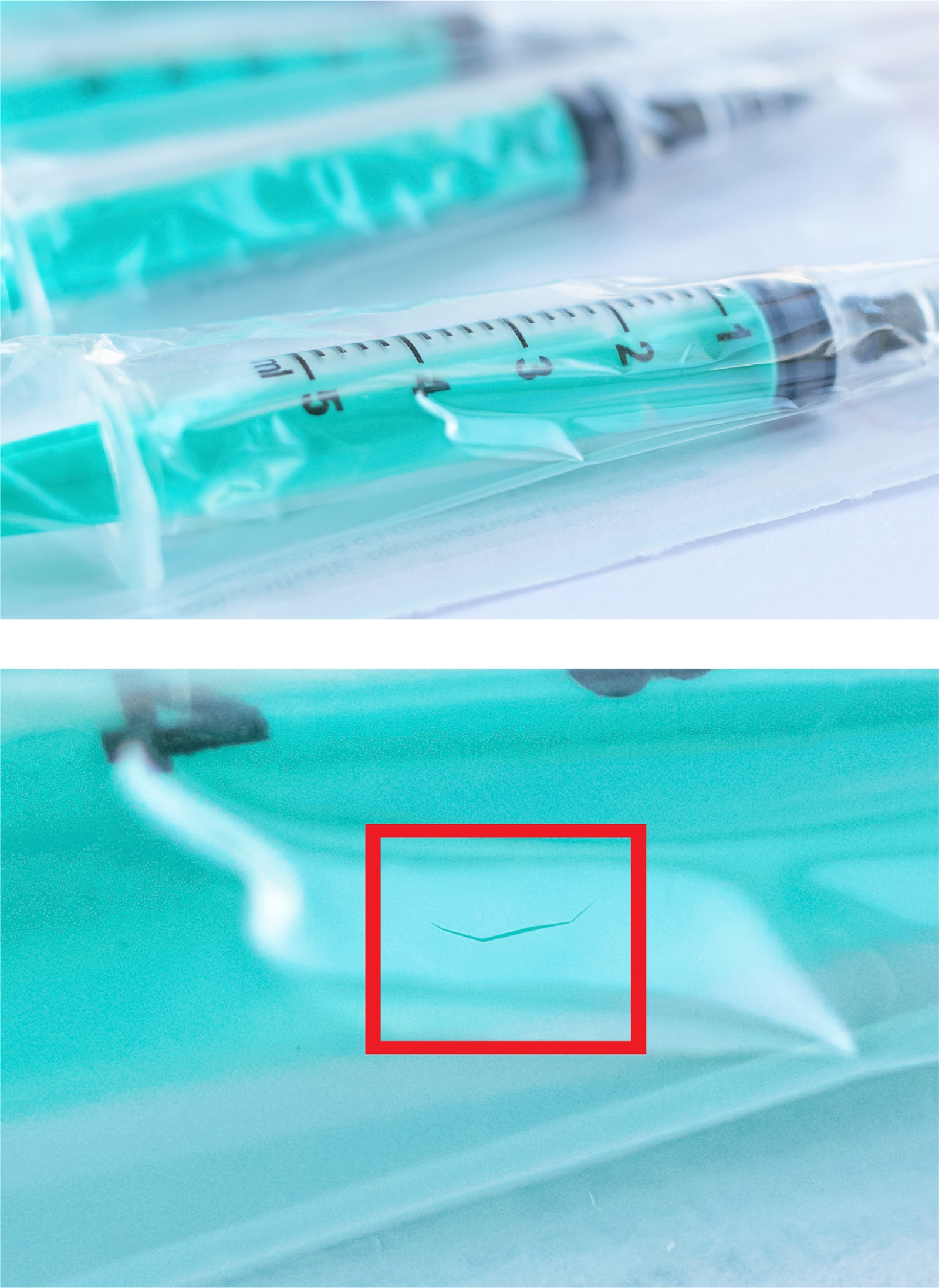
- Vial Contamination InspectionVials and syringes must be thoroughly inspected prior to being sent out, as contaminants in these containers can mean the difference between life and death. Vials and syringes must also be inspected for defects such as cracks to avoid potential safety problems, not to mention recalls, lost revenue, and damaged customer relationships. Machine vision technology can help with vial and syringe inspection, but rules-based algorithms might struggle with the various defect types and false positives caused by issues like transparency and reflections. LandingLens software allows teams to build reliable AI models, automating processes that were previously considered impossible to automate.
- Medical Device Seal InspectionWhether it’s a surgical tool, ventilator, or heart valve, medical devices must be sealed into sterile packaging before being shipped off to the customer. Failure to do so presents risks to patient safety, company reputation, and the bottom line. While traditional machine vision technology can provide an automated means for inspection, deep learning software can help in scenarios that prove difficult for rules-based techniques. These include identifying contamination and improper seals — problems that could lead to drastic outcomes if the product escapes the manufacturing plant floor.
- Medical Device PCB InspectionPrinted circuit boards used in medical devices must be thoroughly inspected, but conducting mode and effects analysis for each wafer, pin, joint, and environment change is difficult. With rules-based machine vision, defining defects such as tilts, burns, foreign material presence, voids, and chips can be difficult or impossible. Deep learning software, however, allows manufacturers to detect and classify these defects.
- Final Assembly VerificationManufacturers must perform final inspections of medical devices and other products to ensure that they are properly assembled and will work once they reach the customer. Tasks include locating, inspecting, and identifying manufactured components. Such tasks are often performed with machine vision systems, but factors such as lighting variations, random part or product orientation, and the sheer number of defect types might lead companies to manually perform this inspection. Deep learning, however, offers a reliable method for automated assembly verification.
- Biomedical ApplicationsDeep learning can be applied in biomedical research applications, such as identifying the number, location, and type of cells in images. Deep learning software can also be applied to other scenarios, including disease detection and diagnosis, drug discovery research, clinical trials, and pattern recognition for genomics.



LandingLens Benefits for Pharmaceutical Inspection
LandingLens, an industry-first data-centric artificial intelligence (AI) visual inspection platform, helps improve inspection accuracy and reduce false positives. The end-to-end platform standardizes deep learning solutions that reduce development time and scale projects easily to multiple facilities across the globe.
Meet Strict Regulations, Keep Compliant
Medical and pharmaceutical companies must follow strict regulations from agencies such as the Food and Drug Administration. Maintaining product quality is key to complying with these regulations. LandingLens deep learning software can help companies stay compliant by improving inspection accuracy and catching defects and irregularities early in the process. Manufacturers in this industry are also subject to process validations which help demonstrate that the manufacturing process will consistently produce conforming products. LandingLens can help support the validation process by maintaining clean, thorough data and results.


Achieve Zero Product Escapes
There’s no room for error when it comes to manufacturing environments, and this is especially true for medical and pharmaceutical manufacturing. Companies need machine vision and deep learning software systems to prevent defective products from leaving the factory floor. This is crucial for avoiding corrective actions, increasing productivity, delivering quality products, and maintaining solid customer relationships.