Computer Vision inMedical Devices
In the world of medical technology, there’s no room for compromise on safety, effectiveness, and quality. Computer vision powered by AI helps to maintain the highest standards for inspection and defect detection at the manufacturing stage, to ensure the highest possible device performance and reliability.

Applications of AI for Medical Devices
LandingLens offers end-to-end computer vision and deep learning for augmenting medical device inspection processes. The LandingLens platform simplifies deep-learning development and offers an efficient, intuitive interface that enables very high accuracy and throughput for quality control inspections.
Medical Device Inspection
While several inspection tasks in medical and pharmaceutical settings can be done manually, the inspection of nano-scale components is not one of them. Manufacturers must deploy imaging technology to inspect for defects and abnormalities, oftentimes down to the micron level. This involves manual inspection with a microscope, which includes multiple steps and can be susceptible to human errors and inconsistency. Deep learning software can automatically process microscopy images and classify defects or irregularities.

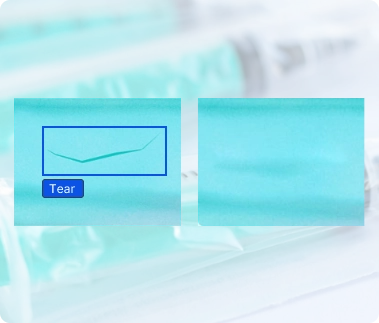
Medical Device Seal Inspection
For items such as surgical tools, heart valves, ventilators, and stents that are sealed in sterile packaging before shipping, LandingLens enhances detection of contamination and improper or incomplete seals. LandingLens augments traditional rules-based inspection techniques with deep learning AI models, improving accuracy and helping to increase output quality
Medical Device PCB Inspection
Performing mode and effects analyses for each wafer, pin, joint, and environment change on printed circuit boards (PCBs) is no easy task. LandingLens brings sophisticated AI-powered deep learning capabilities to enhance PCB inspection. The LandingLens platform can pinpoint defects including tilts, burns, foreign-material presence, voids, and a host of exceptions, helping to ensure ground-up component quality at every manufacturing stage.


Final Assembly Verification
Manufacturers must perform final inspections of medical devices and other products to ensure that they are properly assembled and will work once they reach the customer. Tasks include locating, inspecting, and identifying manufactured components. Such tasks are often performed with machine vision systems, but factors such as lighting variations, random part or product orientation, and the sheer number of defect types might lead companies to manually perform this inspection. Deep learning, however, offers a reliable method for automated assembly verification.
LandingLens Benefits for Computer Vision in Medical Devices and Technology
Medical device manufacturing and production processes require inspection to ensure quality and reliability. LandingLens takes visual examination to the next level by adding AI-powered deep learning, combined with an intuitive user interface. When it comes to AI in medical devices and technology, the LandingLens platform elevates inspection accuracy, highlighting potential defects, providing an added layer of quality management all while boosting productivity, throughput, and profitability. Explore the transformative advantages of computer vision in medical technology and healthcare.

Ease Regulatory
Compliance
- Medical -device manufacturers face strict regulations and guidelines that leave no room for error. LandingLens makes it easier for companies to achieve compliance by supporting extremely accurate visual inspections of defects, and providing a documented audit trail of those inspections.
- With LandingLens, manufacturers can improve quality control, in turn helping to achieve regulatory compliance and – ultimately – enhance patient outcomes.

Improve Inspection Accuracy
and Quality
- The computer vision capabilities from LandingLens helps medical device manufacturers to identify defects more reliably and earlier during the production process. By catching possible defects and enhancing build quality, manufacturers can reduce the likelihood of costly product recalls, and protect brand reputation.
- Further, increased production quality enabled by LandingLens will translate to improved device reliability, and contribute to superior clinical performance.
AI Medical Devices & Technology Resources
Improve Medical-Device Inspection Accuracy with Deep Learn

How to Set Up for Success in Deep Learning Development




